http://www.rhci-online.net/radiogram/radiogram.htm
RSID: <<2020-05-17T01:30Z MFSK-64 @
http://www.rhci-online.net/radiogram/radiogram.htm
RSID: <<2020-05-17T01:30Z MFSK-64 @
9925000+1500>>
Saxophonist Alto Reed was born May
16, 1948.
Sending Pic:164x231;

altoreed.com (?)
====>
https://web.archive.org/web/20160324192442/http://altoreed.com/about/
altoreedpresents.com
Please report your decode to
themightykbc@gmail.com

RSID: <<2020-05-14T23:31Z
MFSK-32 @
9265000+1500>>
Welcome to program 152 of Shortwave Radiogram.
I'm Kim Andrew Elliott in Arlington, Virginia USA.
Here is the lineup for today's program, in MFSK modes as noted:
1:39 MFSK32: Program preview (now)
2:44 New solar cells work well indoors*
8:18 MFSK54: COVID-19 drug remdesivir is here. Now what?
[typo: "64"]
13:38 This week's images*
28:10 MFSK32: Closing announcements
* with image(s)
Please send reception reports to
radiogram@verizon.net
And visit http://swradiogram.net
Twitter:
@SWRadiogram
From New Atlas:
Indoor perovskite solar cells pack high efficiency in artificial
light
Michael Irving
11 May 2020
It might sound counterintuitive to put solar cells indoors, but
there's still plenty of light energy inside that's largely going
to waste. Now, a team of engineers is claiming to have created
flexible perovskite solar cells with the highest efficiency of
any indoor flexible solar cells.
In homes, stores, offices and other buildings, interior lighting
generally shines at between 100 and 500 lux. It's obviously a far
cry short of sunlight, which typically bathes the world in about
110,000 lux, but it's still enough to work with.
The new design comes from researchers at Tor Vergata University
of Rome, Universidad Surcolombiana, and the Fraunhofer Institute,
and involves depositing perovskite solar cells onto glass
substrates that are ultra-thin, flexible and coated in indium tin
oxide (ITO). The end result is photovoltaic cells that are
bendable, strong, and can harvest light at indoor brightnesses.
In tests under LED illumination, the cells were found to have
efficiencies of 20.6 percent under 200 lux, and 22.6 percent
under 400 lux. The team says that this makes them the highest
reported efficiencies of any flexible and curvable indoor
photovoltaic technology
And that does seem to hold true, if not just because of the
several qualifiers included in the claim. Previous indoor
photovoltaic cells we've seen have had efficiencies as low as 10
percent, while those that ranked higher – up to 26.1 percent –
only did so under 1,000 lux.
Power densities on the new designs are relatively low, but that's
to be expected with indoor cells. In this case, under 200 lux the
density reaches 16.7 microWatts per cm2, and 400 lux bumps it up
to 35 microWatts. That means these cells won't be running
anything too power-hungry, but they might be useful for small
sensors or Internet of Things devices.
The researchers also say that the manufacturing method is easy to
scale up, so these flexible solar cells should be relatively
cheap to make in bulk.
The research was published in the journal Cell Reports Physical
Science.
Source: Fraunhofer Institute
https://newatlas.com/energy/indoor-perovskite-solar-cells-high-efficiency-artificial-light/
Image: A sample of the new flexible, perovskite solar cell ...
Sending Pic:168x179C;
Shortwave Radiogram changes to MFSK64 ...
RSID: <<2020-05-14T23:38Z MFSK-64 @ 9265000+1500>>
This is Shortwave Radiogram in MFSK64
Please send your reception report to radiogram@verizon.net
From Science News:
The new COVID-19 drug remdesivir is here. Now what?
Short supplies and limits on who qualifies mean more options
are needed to end the pandemic
Tina Hesman Saey
13 May 2020
Though remdesivir, a new treatment for COVID-19, has been hailed
as a game changer, most people sick with the coronavirus will
have to recover or die without getting the drug.
"Everyone won't be able to get it, because there just isn't
enough of it at this point in time," says Raymond Woosley, a
cardiologist and clinical pharmacologist at the University of
Arizona College of Medicine Phoenix.
Supplies are limited and the federal government is asking state
health departments to distribute the drug to hospitals treating
COVID-19 patients. Vials of the still-experimental medication
have been distributed to 13 states so far. But the Infectious
Diseases Society of America has warned that tens of thousands of
people each month may need the treatment throughout the summer.
Remdesivir shortened recovery time for seriously ill patients by
four days in a clinical trial comparing the drug with a placebo.
Those results were considered so promising that a safety
oversight committee stopped the clinical trial early to give
people taking the placebo a chance to get the drug. Remdesivir
will become the standard of care for the coronavirus, Anthony
Fauci, director of the U.S. National Institute of Allergy and
Infectious Diseases, predicted at a news conference at the White
House on April 29 announcing the results. On May 1, the U.S. Food
and Drug Administration issued an emergency use authorization,
allowing remdesivir to be used for hospitalized people with
confirmed cases of COVID-19, whose blood oxygen levels fall to 94
percent or lower.
Remdesivir is an antiviral drug that works by mimicking building
blocks of the coronavirus's genetic material, RNA. As the virus
copies its RNA, remdesivir takes the place of some building
blocks, stopping or slowing viral replication. In laboratory and
animal tests, remdesivir has been effective against a wide
variety of coronaviruses, and human trials showed that it helped
some people survive Ebola). Now, the new trial suggests the drug
can also fight SARS-CoV-2 infections.
"While the remdesivir news is positive - anything that is helping
patients is positive - this is not the coronavirus cure for
everyone," says Esther Krofah, executive director of FasterCures,
a Washington, D.C.-based nonprofit that's part of the Milken
Institute think tank. The center is tracking hundreds of drugs
and vaccines being tested against the coronavirus. Because the
drug is given intravenously, only hospitalized patients can get
it. "If you're moderately ill or you're showing very mild
symptoms, it would continue to be the case that you would
quarantine at home and monitor your symptoms."
Limited supplies of remdesivir can stretch only so far to meet
great demand.
Gilead Sciences, the maker of remdesivir, has donated its entire
supply of the drug, which could treat about 140,000 people for 10
days each. The company, based in Foster City, Calif., ran its own
clinical trial and found that treating for five days may be
enough, potentially stretching the drug's supply to more than
250,000 people worldwide. The company anticipates scaling up
production to make more than 1 million treatment courses of
remdesivir by year's end. That's based on using the 10-day
treatment regimen. Using a shorter course might mean 2 million
people could access the drug this year.
"Gilead has committed to manufacturing however many doses are
needed. But it's a complex drug from a manufacturing
perspective," Krofah says. The company warned in a May 5
statement that making remdesivir requires scarce raw materials,
and disruptions to supply chains for those materials might slow
production or limit the amount of remdesivir that can be produce.
Lessons from trials
In addition, it's not at all clear that remdesivir is the best
treatment against SARS-CoV-2, some scientists say, and its
widespread use might even make finding better treatments harder.
A proven treatment is welcome, but stopping the pandemic will
require more than remdesivir, experts agree.
A clinical trial conducted in Wuhan, China, did not find any
statistically meaningful difference in recovery for people
getting remdesivir infusions compared with people receiving a
placebo. Still, the larger study conducted by Fauci's institute
indicates remdesivir may really help people get better faster.
Because it followed more patients, the larger trial probably is
closer to getting the right answer, researchers say. But
scientists are concerned that the trial data are not yet
available for review.
Another problem is that the trial twice changed the measure by
which it judged the drug's success. That's usually a warning
sign. In general, "if you change the goal posts it makes you
wonder … if this is a real effect, or just one of many that they
chose to be the outcome, ignoring the one the study was
originally designed for," Woosley says.
Initially, the trial was supposed to measure improvement on day
14 after treatment began. Once it became clear that COVID-19 is a
lingering illness, the researchers changed the judgment date. But
they also realized that picking just one date may have problems
of its own, the institute said in a statement. While the trial
was still under way and before anyone knew which patients were
getting remedesivir or a placebo, "NIAID statisticians performed
modeling of what happens if the right day is not picked for
assessment, which revealed that meaningful treatment effects
could be missed with that primary endpoint," the statement
explains. On April 16, the outcome goal was changed to gauge an
improvement in recovery time. "The change in primary endpoint
seemed appropriate given the evolving clinical data," the NIAID
statement says.
But that still leaves researchers with many questions about
remdesivir, says William Powderly, an infectious disease doctor
at Washington University School of Medicine in St. Louis. Among
those questions are how effective the drug really is, who are the
patients that will benefit most from the treatment, when is the
best time to give the drug, and what are the side effects,
Powderly says. "We have a drug that appears to have an effect. Is
it a slam dunk? Is it a home run? No."
Remdesivir did not have a statistically meaningful effect on
preventing death. In the NIAID trial, 8 percent of people on
remdesivir died, while 11 percent in the placebo group died.
"It's great to see progress," Powderly says, "but what we would
really like to see is a much more dramatic effect on survival."
...
Full text:
https://www.sciencenews.org/article/coronavirus-covid-19-drug-remdesivir-supply-chain
This is Shortwave Radiogram in MFSK64
Please send your reception report to radiogram@verizon.net
This week's images ...
A deliveryman for a wine shop, and his dog, in London. From
bit.ly/2WUmohF ...
https://www.de24.news/2020/05/coronavirus-uk-plaene-zur-lockerung-der-sperrung-in-zweiwoechentlichen-schritten.html
Sending Pic:187x183C;
Big Four Bridge, a former railroad bridge, now a pedestrian
crossing that connects Louisville, Kentucky and Jeffersonville,
Indiana, across the Ohio River. From
bit.ly/2y4z2T2 ...
Sending Pic:211x135C;

"Guardians of the Giraffe" (Kenya), one of the winners of the
BigPicture nature photography contest. From
bit.ly/3bpDTM8 ...
Sending Pic:207x147C;

A spring view of Paisley, Scotland. From
bbc.in/3fO262c ...
Sending Pic:171x162C;
A European robin in Cumbernauld, Scotland. From
bbc.in/3fO262c
...
Sending Pic:170x168C;

Sunset in Aberdeen, Scotland, from the Torry Battery. From
bbc.in/3fO262c ...
Sending Pic:207x135C;

Sunlit buttercups along Rock Creek Trail in Washington DC. From
wapo.st/3fPAqKn ...
Sending Pic:205x148C;

Our art of the week is from the "You Hear the Colours" series by
Angela Johal. From
bit.ly/3dKw8lE ...
Sending Pic:209x167C;

Shortwave Radiogram returns to MFSK32 ...
-
RSID: <<2020-05-14T23:58Z MFSK-32 @ 9265000+1500>>
This is Shortwave Radiogram in MFSK32 ...
Shortwave Radiogram is transmitted by:
WRMI, Radio Miami International, wrmi.net
and
WINB Shortwave, winb.com
Please send reception reports to
radiogram@verizon.net
And visit http://swradiogram.net
Twitter:
@SWRadiogram or
twitter.com/swradiogram
I'm Kim Elliott. Please join us for the next Shortwave
Radiogram.
|
Ending music: https://www.midomi.com/index.php?action=main.track&track_id=100868269594851881&from=voice_search
Little Richard - Wonderin'
[Take 1]
|
http://www.rhci-online.net/radiogram/radiogram.htm
|
QTH: |
D-06193 Petersberg (Germany/Germania) |
|
|
Ant.: |
Dipol for 40m-Band & Boomerang Antenna 11m-Band |
|
|
RX for RF: |
FRG-100B + IF-mixer & ICOM IC-R75 + IF-mixer |
|
|
Software IF: |
con STUDIO1 - Software italiano per SDR [S-AM-USB/LSB] + beta 11 Version 2.80 (August 21, 2018) - for scheduled IF-recording |
|
|
Software AF: |
Fldigi-4.0.18 + flmsg-4.0.7 images-fldigifiles on homedrive.lnk |
|
|
OS: |
German XP-SP3 with support for asian languages |
German W7 32bit + 64bit |
|
PC: |
MEDION Titanium 8008 (since 2003) [ P4 - 2,6 GHz] |
MSI-CR70-2MP345W7 (since2014) [i5 -P3560 ( 2 x 2,6GHz) ] |
http://wiki.radioreference.com/index.php/Decoding_the_SW_Radiogram_Broadcasts
https://www.qsl.net/ve7vv/Files/Digital%20Modes.pdf
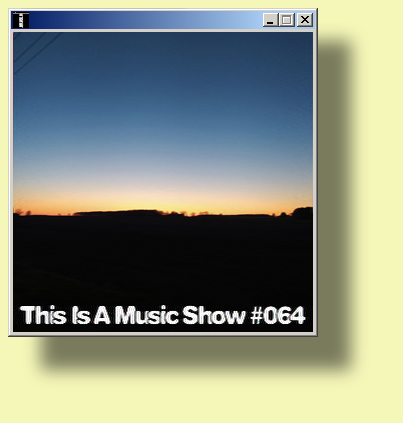
2020-05-13 2100-2200z 7780 kHz WRMI TIAMS#064 (1st)
2020-05-14 0200-0300z 5850 kHz WRMI TIAMS#064 (2nd)
2020-05-14 2330-2400z 9265 kHz WINB SWRG#152 (1st)
2020-05-15 1000-1030z 6070 kHz ch292 RNEI#04
2020-05-15 1030-1100z 6070 kHz ch292 TIAEMS-May-2020
2020-05-15 1000-1100z 3210 kHz Unique Radio Australia TIAMS#064 (3rd)

RSID: <<2020-05-1510:47Z MFSK-64 @
3210000+1500>>

This Is A Music Show #064
***ALSO*** |
"...18:00 AEST 3210 KHz is in use USB all scheduled nights ..." [AEST = UTC+10h] |
----------------------------------------
PLAYLIST
|
Swirlies - San
Cristobal de las Casas |
- - - |
|
|
Yeah Yeah
Yeahs - Bang ♫♥ |
https://www.midomi.com/index.php?action=main.track&track_id=100206362884026021&from=voice_search https://www.midomi.com/index.php?action=main.track&track_id=100081751662306488&from=voice_search - - -
|
|
|
Perfume -
ポリリズム
[Polyrhythm] |
https://www.midomi.com/index.php?action=main.track&track_id=100592781214509159&from=voice_search - - - https://www.midomi.com/index.php?action=main.track&track_id=100721429837867617&from=voice_search
|
|
|
Space Explosion - Space Explosion
(1997) -----
|
- - -
Feat.:
Dieter Moebius (CLUSTER), Mani
Neumeier (GURU GURU), Jürgen Engler (DIE KRUPPS),
Chris Karrer (AMON DÜÜL,
Werner Diermaier (FAUST) and Jean-Hervé Peron (FAUST) |
|
|
|
|
|
|
The Remote
Viewer - It Occured To Me. And Went Away. |
https://www.midomi.com/index.php?action=main.track&track_id=100832452357746660&from=voice_search
|
|
Links of note:
Blond Redhead Futurism vs. Passéism
Live in '97
https://youtu.be/l7UmIfILZD0?t=1716
-----
Please send reception reports/comments:
thisisamusicshow@gmail.com
Follow TIAMS on Twitter:
www.twitter.com/ThisIsAMusicSho/
------
Thanks for listening!
--YOUR HOST--
EOM
Sending Pic:300x300Cp4;
Sending Pic:300x300Cp2;

RSID: <<2020-05-15T10:26Z MFSK-32 @ 6070000+1500>>
Thanks for listening to RNEI 4!
qsl@rnei.org.
Played today:
https://www.dropbox.com/s/1slac0ucinwf1zz/2020-05-15_RNEI04-The_Roop-On_Fire_cut.wav?dl=0
TIAMS has made another 30 minute express music show for after our broadcast for you to enjoy!
|
Date |
Time |
Frequency |
Target Zone |
|
Sunday the 3rd of May 2020 |
18:00 UTC |
6070KHz |
Zone 1+2 |
|
Saturday the 9th of May 2020 |
18:00 UTC |
6070KHz |
Zone 1+2 |
|
Friday the 15th of May 2020 |
10:00 UTC |
6070KHz |
Zone 1 |
|
Saturday the 23rd of May 2020 |
00:00 UTC |
6070KHz |
Zone 2+ |
|
Sunday the 31st of May 2020 |
18:00 UTC |
6070KHz |
Zone 1+2 |